An Unknown Element Is A Mixture Of Isotopes
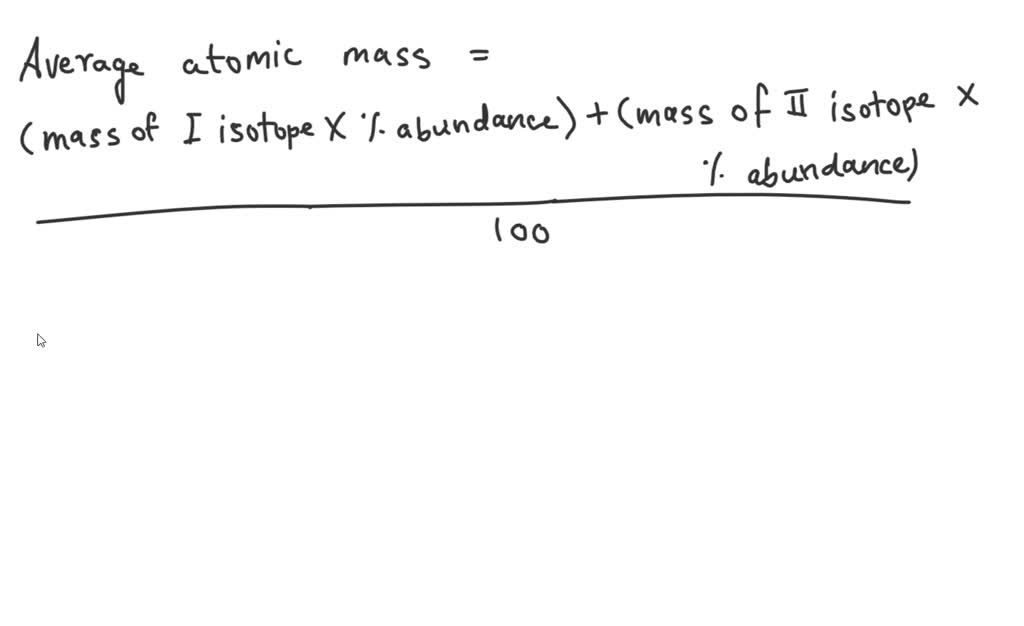
Imagine your favorite ice cream. You probably think of it as one thing, right? Chocolate, vanilla, strawberry – a perfect, unified flavor. But what if I told you that even that delicious scoop is actually a tiny bit of a secret party, with different versions of "chocolate" all hanging out together?
Well, that's kind of how it is with the fundamental building blocks of everything around us, the things we call elements. You've heard of gold, iron, or maybe even the fizz in your soda, carbon. We often think of them as these neat, distinct characters in the grand play of the universe.
But here's the super cool, slightly mind-bending secret: most of these elements aren't just one thing. They're actually a happy jumble, a collection of "cousins" that are all very similar but have tiny, almost invisible differences. These cousins are called isotopes.
Must Read
Think of it like a family reunion. You've got Aunt Carol, who's always telling jokes, and Uncle Bob, who loves to tell stories. They look alike, share a last name, and you can tell they're related, but they have their own unique quirks and personalities.
In the world of atoms, the "cousins" (isotopes) of an element share the same number of important bits called protons, which is what makes them that specific element. It’s like having the same family crest. But they might have a different number of other bits called neutrons.
These extra or fewer neutrons are like Uncle Bob having a slightly different hairline than Aunt Carol. It doesn't change who they are fundamentally, but it makes them technically a little different. And this is where the "unknown element" part of our story pops up!

Sometimes, when scientists first discover something new in nature, they might not realize it’s already a mixture. They might see a sample, analyze it, and say, "Wow, this is a brand new element!" They'd give it a cool name, maybe after a favorite scientist or a place they love. It’s like finding a new flavor of cookie and thinking you invented it.
But then, as they study it more closely, they start to notice that this "new element" is behaving a little… odd. It’s not acting like a perfectly uniform substance. It's like if that cookie you "invented" sometimes tasted a little bit like chocolate chips, and other times had hints of caramel, even though you thought it was supposed to be plain sugar.
This is when the investigative part of science kicks in. They'd start to pull apart the "cookie" to see what's inside. And voilà! They discover that their "new element" isn't just one type of atom. It's actually a blend, a mixture of these different isotopes.
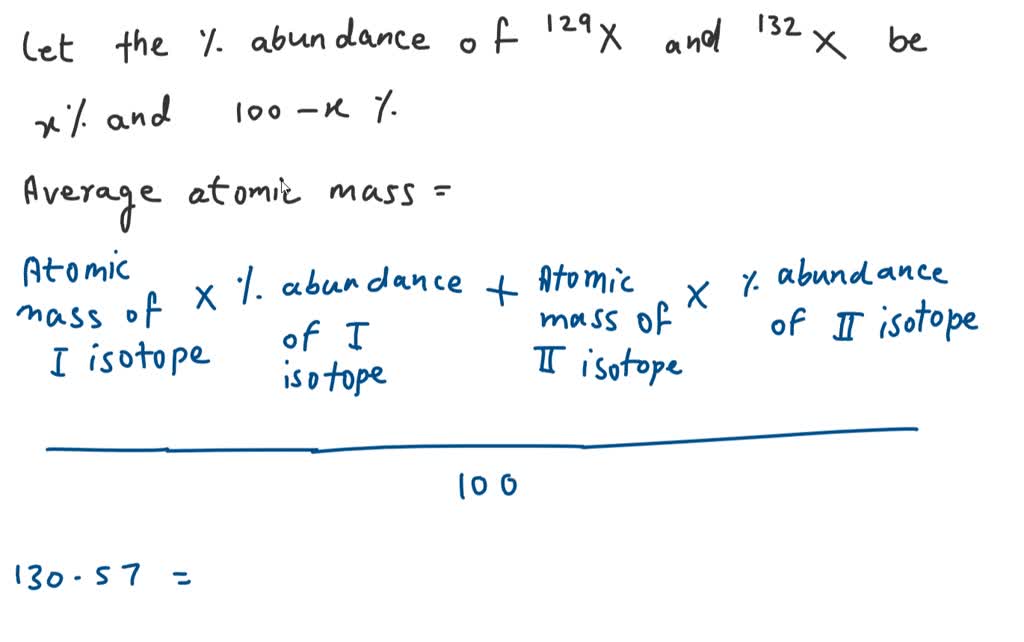
So, what they thought was a brand-new, solo star on the periodic table turned out to be more like a band, with several "versions" of the same element playing together. It’s a little bit like thinking you've discovered a new kind of bird, only to realize it’s a flock of slightly different-colored sparrows all hanging out.
This realization can be both a little embarrassing for the scientists (imagine announcing a discovery that turns out to be a known thing!) and incredibly exciting. It means they've uncovered more complexity and nuance in the universe.
Think about oxygen, the stuff we breathe. We think of it as just oxygen. But oxygen actually comes in a few isotopic forms. The most common one is like the reliable, everyday version. But there are rarer, heavier cousins out there too.
Sometimes, these isotopic differences can have really important consequences. For example, in dating ancient artifacts, scientists use isotopes of carbon (carbon-14) to figure out how old things are. This isotope decays at a predictable rate, acting like a tiny, natural clock.

If a scientist were to discover a new type of ancient pottery, and they analyzed its carbon content, they'd be relying on the fact that carbon has these isotopic forms to tell them its age. Without that isotopic "mix," the clock wouldn't work!
It’s also a bit heartwarming, in a way. It shows that even the most basic things are made up of these intricate, interconnected families. Nothing in the universe is truly alone; everything is part of a larger, often surprising, community.
So, the next time you look at a piece of metal, or even a breath of air, remember that you're not just seeing a single, simple element. You're looking at a bustling gathering, a party of isotopes all playing their part. It's a reminder that the world around us is far more interesting and diverse than we often imagine, even down to the tiniest of particles.
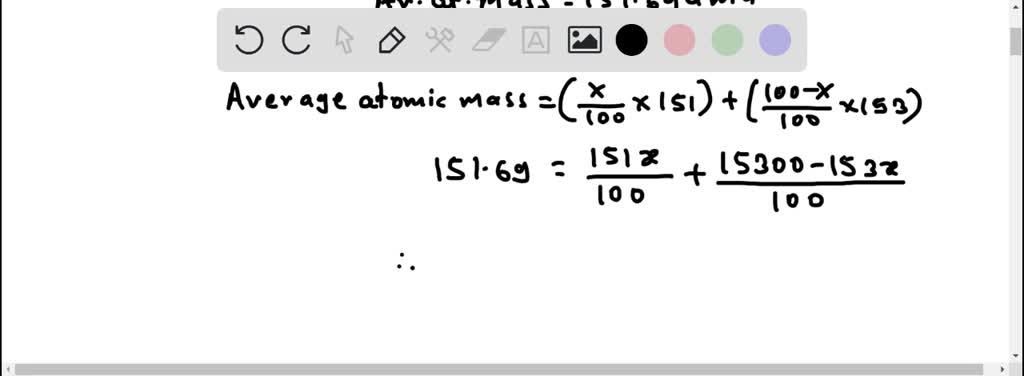
This discovery process, where an "unknown" turns out to be a mixture, has happened many times in the history of chemistry. It’s a testament to how much there is still to learn, and how often nature likes to surprise us with its cleverness.
It's like finding out your favorite superhero has a whole team of sidekicks you never knew about, each with their own special powers, all working together. That's the essence of isotopes within an element – a team effort!
So, while we might give elements simple names on the periodic table, the reality is a beautiful, often hidden, complexity. It’s a story of discovery, a touch of surprise, and a whole lot of interconnectedness in the fabric of our universe.
And that, my friends, is the fun secret life of elements and their ever-present, slightly different, isotopic families!





