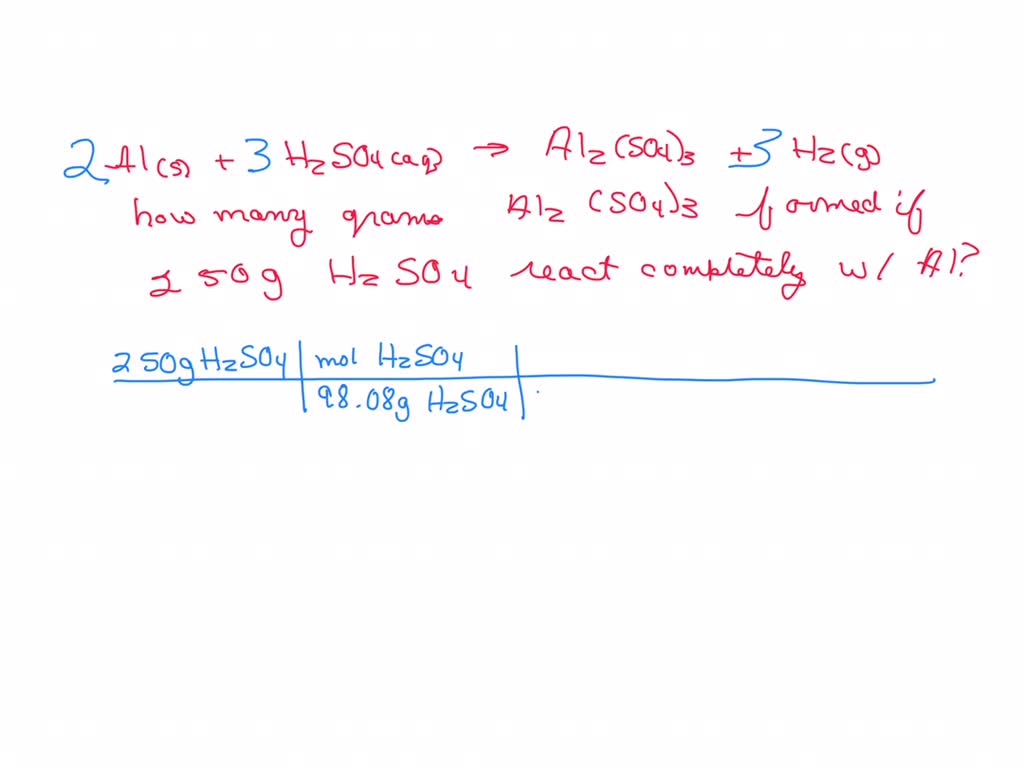
Aluminum Metal Reacts With Hydrochloric Acid

Hey there, science curious folks! Ever just stare at stuff and wonder what makes it tick? Like, what happens if you toss something shiny and metallic into a fizzy liquid? Today, we're diving into a little kitchen-counter-science experiment, or at least the idea of one, that's pretty darn cool: aluminum metal reacting with hydrochloric acid. Sounds a bit like a mad scientist’s dream, right? But it’s actually way more accessible and, dare I say, fascinating than you might think.
So, what are we even talking about here? We've got aluminum, that lightweight, shiny metal we see everywhere – think soda cans, foil, even some window frames. And then we have hydrochloric acid, a strong acid that’s, well, acidic. You might have heard of it being used in industrial cleaning or even, in very diluted forms, in our own stomachs to help digest food. (Don't try mixing these at home, though! Safety first, always.)
When these two buddies meet, things get interesting. It’s not just a gentle handshake; it’s more like a fizzy, bubbly dance. Imagine throwing a tiny, crumpled-up aluminum foil ball into a glass of soda. You’d expect some bubbles, right? Well, with hydrochloric acid, it’s kind of like that, but on steroids. You start seeing this rapid production of tiny bubbles. What are these bubbles, you ask? They’re actually hydrogen gas!
Must Read
Think of it like this: the aluminum atoms are like little kids who are a bit restless. The hydrochloric acid is like a really energetic grown-up who says, "Okay, kids, let's play!" The acid basically helps the aluminum atoms get rid of some of their buddies (electrons, in chemistry talk) and team up with parts of the acid instead. And as a byproduct of this energetic game, hydrogen atoms, which were also hanging out in the acid, decide to buddy up and form hydrogen gas. Poof! Bubbles!
So, the aluminum metal, which was just sitting there being all metallic and solid, starts to get dissolved. It’s transforming into something new. This process is called a chemical reaction. It’s like taking Lego bricks and rearranging them to build something completely different. The original aluminum is gone, and in its place, we get new substances – the dissolved aluminum compounds and, of course, that lively hydrogen gas.
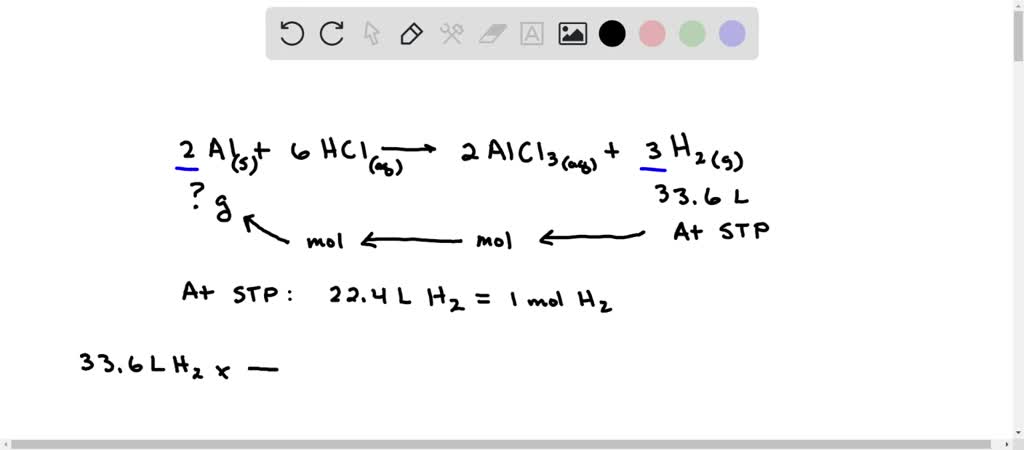
Why is this reaction so cool?
Beyond the obvious fizz and pop, there are several reasons why this interaction between aluminum and hydrochloric acid is neat. For starters, it’s a classic example of how different substances can interact to create entirely new ones. It’s the fundamental idea behind chemistry – taking the ordinary and making the extraordinary.
And the speed of it! Depending on how concentrated the acid is and how finely divided the aluminum is (think powder versus a big sheet), this reaction can happen really fast. It’s like watching a time-lapse video of a flower blooming, but in reverse, where something solid just seems to vanish into thin air, leaving behind a trail of energetic gas.

Plus, let’s not forget the science behind it. Aluminum is a pretty reactive metal, but it also has this clever trick up its sleeve. When exposed to air, it forms a thin, invisible layer of aluminum oxide. This layer is like a protective shield, preventing the aluminum underneath from reacting too easily with things like oxygen or even water. However, hydrochloric acid is strong enough to break through this protective shield. Once the shield is down, the real fun begins!
It's a bit like that moment when you finally get through to a busy person on the phone. The oxide layer is the busy signal, and the hydrochloric acid is your persistent calling. Once you’re connected, the conversation (the reaction) can really get going.

Real-world connections (without the drama!)
While you probably shouldn't be mixing these at home, understanding this reaction helps us appreciate things in the real world. For instance, it highlights why certain metals are used in different environments. Aluminum is used in aircraft because it’s lightweight and strong, but engineers have to consider its potential reactions. Hydrochloric acid, on the other hand, has its uses, but it requires careful handling.
It also sheds light on corrosion. While this reaction is a bit more vigorous, the underlying principles of metals reacting with acids are related to how metals can degrade over time in certain conditions. It’s a reminder that even seemingly inert materials are constantly interacting with their surroundings on a molecular level.

Think about cleaning. If you've ever seen a product that claims to remove rust or mineral deposits, there's a good chance it contains some form of acid. These acids are designed to react with and break down unwanted substances, much like hydrochloric acid breaks down aluminum. It’s chemistry at work, making our lives a little cleaner and brighter.
And then there's the sheer wonder of it. The fact that a solid metal can disappear into a liquid, releasing a gas that we can’t even see, is pretty mind-boggling when you stop and think about it. It’s a visual representation of the invisible forces and transformations happening all around us, all the time. It makes you wonder what else is happening in that glass of water you’re drinking, or in the soil beneath your feet.
So, next time you see a shiny aluminum can or hear about acids being used for something, you might just remember this bubbly, energetic reaction. It’s a simple, yet profound, example of how the world works, and it’s pretty darn cool to know that even the most ordinary-looking materials have these extraordinary secrets waiting to be revealed. Keep that curiosity buzzing, folks!