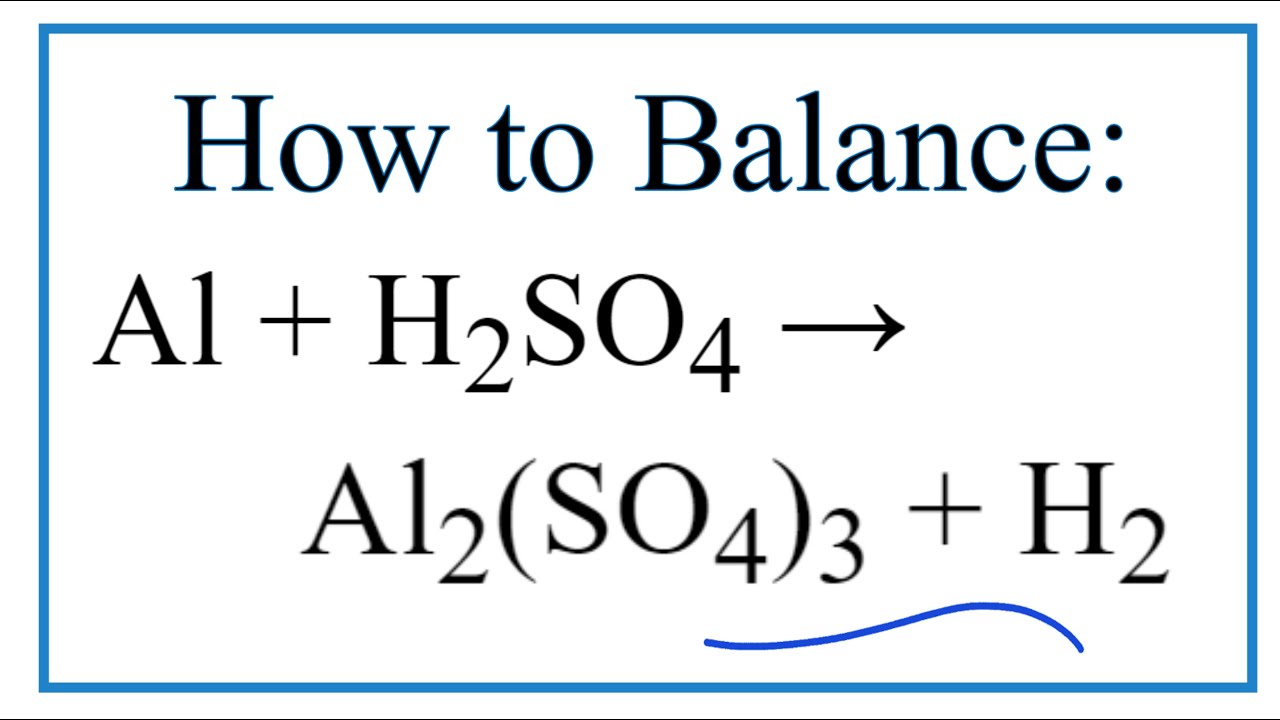
Aluminum And Sulfuric Acid Balanced Equation
Ever wondered what happens when you mix everyday things, and if there’s a way to predict the outcome? Well, one such fascinating interaction involves aluminum and sulfuric acid. It might sound a bit like a science experiment from a high school lab, and in a way, it is! But understanding this particular chemical reaction, and specifically its balanced equation, can be surprisingly relevant and even a little fun. It’s like learning a secret code that tells us exactly how different substances behave when they meet.
So, what’s the big deal about the balanced equation for aluminum and sulfuric acid? Think of it as a chemical recipe that ensures we're not creating or destroying any atoms during a reaction. This principle, known as the Law of Conservation of Mass, is fundamental in chemistry. The balanced equation for the reaction between aluminum (Al) and sulfuric acid (H₂SO₄) is: 2Al + 3H₂SO₄ → Al₂(SO₄)₃ + 3H₂. This equation tells us that two atoms of aluminum react with three molecules of sulfuric acid to produce one molecule of aluminum sulfate and three molecules of hydrogen gas. The magic here is that the number of atoms of each element is the same on both sides of the arrow. No atoms are lost, and no new ones are magically conjured up. This precision is crucial for predicting the amounts of reactants needed and the amounts of products formed in any chemical process.
The benefits of understanding this balanced equation extend beyond just a classroom exercise. In science and industry, it's about efficiency and safety. Knowing the exact proportions means we can optimize industrial processes, ensuring we don't waste expensive materials or produce unwanted byproducts. It also helps us understand potential hazards. For instance, the production of hydrogen gas (H₂) in this reaction is noteworthy. Hydrogen is highly flammable, so knowing this equation helps engineers design safe systems for handling such reactions. In a more everyday context, while you’re unlikely to be mixing these two directly at home, the principles of balanced equations are at play in everything from the way your car's catalytic converter cleans exhaust fumes to how batteries generate electricity.
Must Read
For those curious to explore this a bit further, you don't need a full chemistry set. You can start by simply looking up videos of the reaction online. Seeing it happen, even vicariously, can be incredibly illuminating. You might also explore other common balanced equations, like the burning of natural gas (methane): CH₄ + 2O₂ → CO₂ + 2H₂O. This is a much more accessible example that illustrates the same core concept. If you're in school, pay close attention in your chemistry classes when balancing equations are taught – it’s a skill that unlocks so much understanding. Even just observing everyday products and thinking about the chemical reactions that create them can spark curiosity. The world around us is a constant laboratory, and balanced equations are the keys to understanding its fascinating experiments.