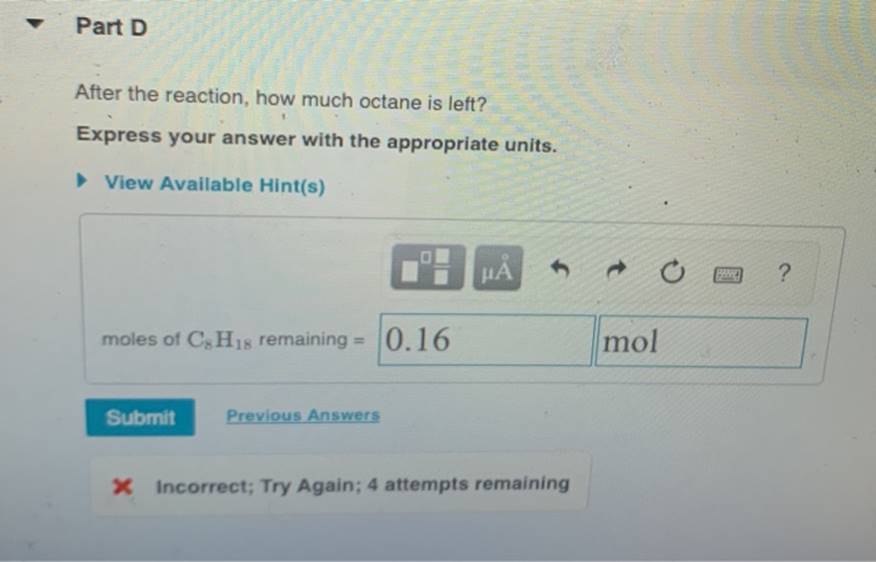
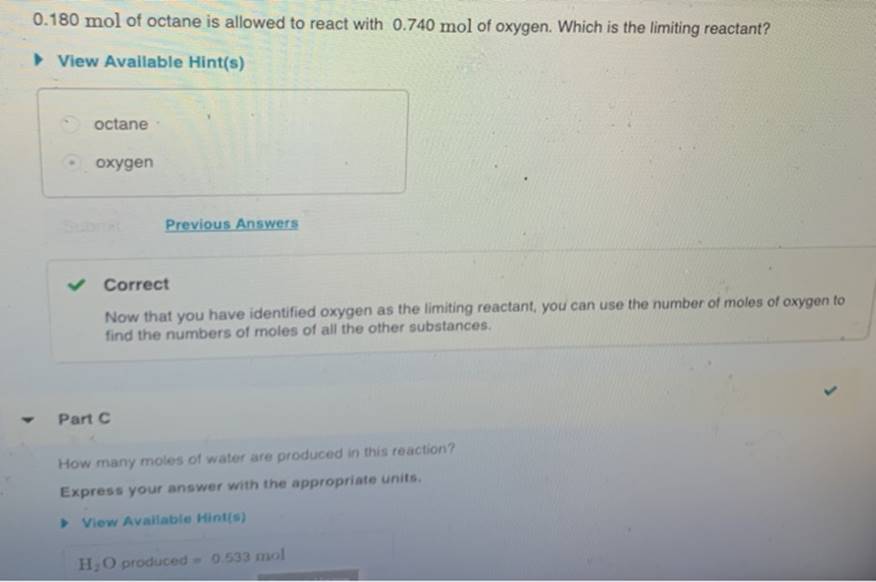
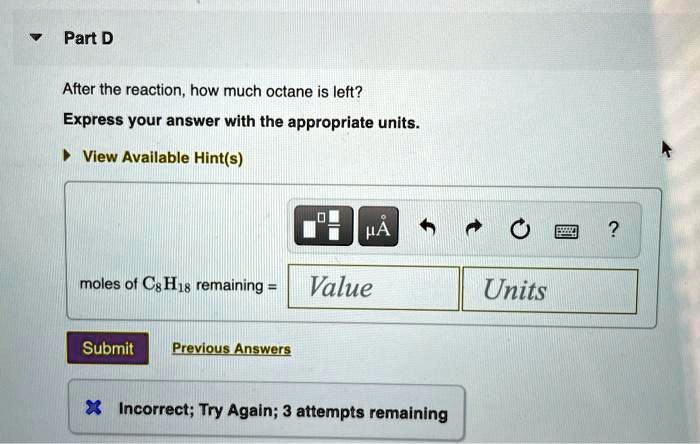
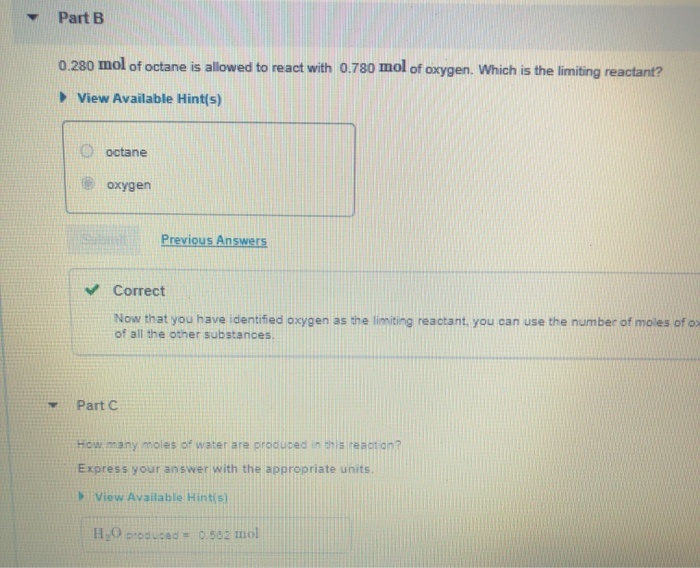
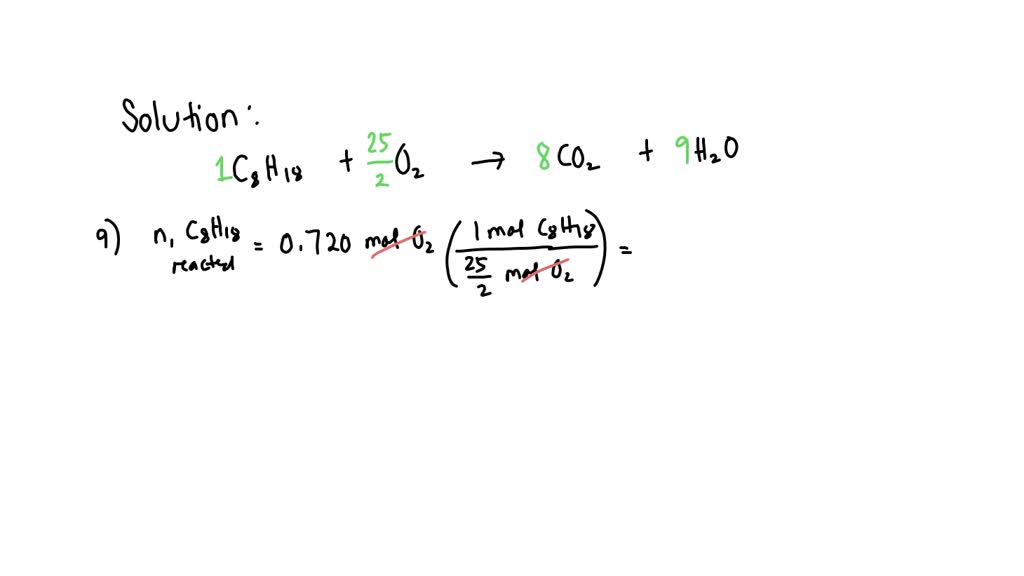After The Reaction How Much Octane Is Left

Ever have one of those days where you feel like you've just run a marathon, even though you only walked to the mailbox? Or maybe you've seen a really exciting movie that left you buzzing for hours afterward? Well, get ready, because we're about to dive into a similar kind of "aftermath" – but this time, it's all about what happens inside your car's engine after it's done its important job of turning fuel into go-power.
We're talking about octane, that magical number you see on the gas pump. You know, like 87, 89, or that fancy 91 or 93 stuff. We often just pick one without thinking too much, like choosing between vanilla and chocolate ice cream. But have you ever wondered what's really going on in there? And more importantly, after your car has done its thing, what happens to that octane?
Think of octane like the fuel's stamina. It's a measure of how well gasoline can resist something called "knocking" or "pinging." Imagine your engine is a tiny concert hall, and the combustion of fuel is a performance. Knocking is like a bunch of clumsy musicians hitting the wrong notes at the wrong time, creating a jarring, unpleasant sound. High-octane fuel is like having a perfectly rehearsed orchestra – it burns smoothly and powerfully, without any of those jarring disruptions.
Must Read
So, when your car's engine is humming along, it's all about controlled explosions. Little bursts of power that push the pistons and make your wheels turn. The fuel, with its octane rating, is the star of this show. It gets compressed, ignited, and bang – energy is released.
Now, here's the fun part: what's left after the performance? You might think the octane just disappears, like the applause after a concert. And in a way, it does! The octane itself, as a distinct molecule, isn't really "leftover" in the tank. It's been transformed into something else during the combustion process.
Think of it like baking a cake. You start with flour, sugar, eggs, and butter. You mix them, bake them, and voilà – you have a delicious cake! The original ingredients are still in the cake, but they've changed. They've become something new and wonderful. Similarly, the octane molecules in your gasoline undergo a chemical reaction. They break down and combine with oxygen to produce energy, which is what powers your car. They also produce exhaust gases – the stuff that comes out of your tailpipe.
The main "outputs" of this combustion are carbon dioxide (CO2) and water (H2O). So, the octane, along with other components of the gasoline, is essentially burned to create energy and these byproducts. It’s not like you’ll find little octane molecules hiding out in your engine after a drive, like finding a stray M&M at the bottom of a candy bag.
But here’s where it gets interesting, and why you might actually care about this. While the octane itself isn't "left," the effects of using a certain octane fuel can linger, in a good way. And understanding this can save you money and keep your car happier.

Let's go back to our concert analogy. If you use low-octane fuel in an engine that needs high-octane fuel, it's like trying to play a delicate classical piece on a drum set. It's going to be rough, it's going to be noisy, and it's definitely not going to sound good. That "knocking" sound? That's your engine protesting. It’s struggling to manage the fuel's tendency to ignite prematurely. This can lead to decreased performance and, over time, can actually damage your engine's internal parts. Imagine the poor musicians getting stressed and their instruments getting bent out of shape!
On the flip side, if your car is designed for regular, 87-octane fuel, and you keep filling it up with that super-premium 93 stuff, are you really getting a significant benefit? It's like hiring a world-class opera singer to perform a simple nursery rhyme. They can do it, but it's probably overkill, and you're paying a premium for something you don't necessarily need. The engine is designed to work optimally with a certain fuel type. Putting in fuel with a higher octane rating than required usually doesn't give you extra power or better fuel economy. It's like putting extra sprinkles on a cake that's already perfectly frosted – nice, but not essential.
So, "after the reaction," the octane has done its job. It has contributed to the smooth, powerful combustion that moves your car. The real question isn't how much octane is left, but rather, what was the right octane for your car in the first place? And did you use it effectively?
Your car's manufacturer knows best. They’ve designed the engine, and they’ve specified the recommended octane. You can usually find this information in your car's owner's manual, or sometimes even on a sticker inside your fuel door or on the gas cap. It’s like knowing the best way to care for your pet – the breeder or vet has all the insider tips!
Using the recommended octane ensures your engine runs efficiently, minimizes the risk of knocking and potential damage, and often means you're not wasting money on fuel you don't need. It’s about finding that sweet spot, that perfect balance, where your car performs at its best without any unnecessary fuss or expense.
Think of it this way: if you're running a marathon, you need the right kind of shoes. If you're going for a gentle jog in the park, you can wear your comfy trainers. Both have their purpose. Your car is the same. It has its designed "activity" level, and the octane fuel is its specialized gear.
So, the next time you're at the pump, take a moment. Glance at that number. Think about your car. Is it a sporty little number that might appreciate a bit more stamina, or a reliable workhorse that’s perfectly content with its usual fuel? Understanding octane isn't about being a car expert; it's about being a smart car owner.
It’s about ensuring your car's internal concert hall is always filled with harmonious engine sounds, not clunky, off-key notes. And when that happens, you get a smoother ride, a happier engine, and a few extra bucks in your pocket to treat yourself to that vanilla or chocolate ice cream. Now that’s a win-win!