A Voltaic Cell Differs From An Electrolytic

Ever wondered what powers your portable devices, from your phone to your TV remote? It all comes down to a fascinating bit of science called a voltaic cell, also known as a galvanic cell. If you've ever dabbled in chemistry or just enjoy understanding the world around you, you might find yourself intrigued by how we can create electricity from simple materials. But then you hear about something called an electrolytic cell, and things can get a little fuzzy. Don't worry, it's not as complicated as it sounds, and understanding the difference is surprisingly fun and useful!
So, why should you care about the difference between these two types of cells? For starters, it helps demystify everyday technology. For beginners, it’s a great way to grasp fundamental concepts of electricity and chemistry without getting bogged down in jargon. Families can use this as a springboard for fun science experiments at home, turning learning into an engaging activity. And for hobbyists, understanding these principles can open doors to DIY projects, like building small power sources or even exploring more advanced electronics.
The core difference lies in what they do. A voltaic cell is all about generating electricity. Think of it as a little power plant. It takes a spontaneous chemical reaction and converts that chemical energy into electrical energy. Your everyday batteries are the most common example! That AA battery in your remote? That’s a voltaic cell. Even a simple lemon battery you might have made as a kid, using a zinc nail and a copper coin, is a miniature voltaic cell that can light up a tiny LED.
Must Read
On the other hand, an electrolytic cell does the opposite. Instead of generating electricity, it uses electricity to drive a non-spontaneous chemical reaction. It's like a chemist's tool. The most common application you'll see is in electroplating – think of how chrome is added to car parts or how jewelry gets its shiny gold or silver coating. Electricity is used to deposit a thin layer of one metal onto another. Another example is the process of refining metals like aluminum, which requires a huge amount of electrical energy.
To make it even clearer, let's consider a few variations. For voltaic cells, you have different types of batteries, from the simple alkaline ones to more complex rechargeable lithium-ion batteries. For electrolytic cells, besides electroplating, you have electrolysis of water to produce hydrogen and oxygen gas, which is a key area of research for clean energy.
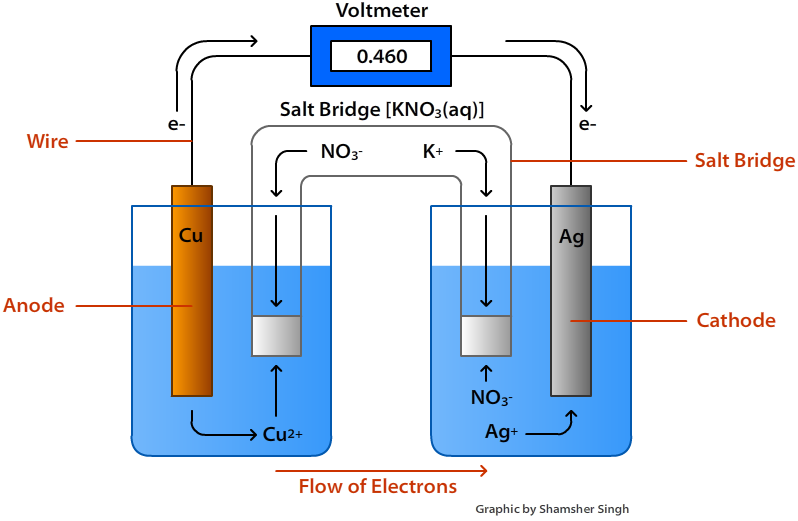
Getting started with understanding this is easy! For a fun, hands-on introduction to voltaic cells, try making a potato battery or a coin battery. You'll need some simple materials like copper and zinc coins, a potato or lemon, and some wires. You can even find kits online that simplify the process. For electrolytic cells, while more complex to set up safely at home, observing videos of electroplating or electrolysis can be a great way to see the principles in action.
Ultimately, understanding the difference between voltaic and electrolytic cells isn't just about memorizing definitions; it's about appreciating the clever ways we harness chemical reactions to power our lives and create new materials. It’s a journey into the heart of how things work, and it’s a journey that can be both incredibly satisfying and surprisingly fun!








