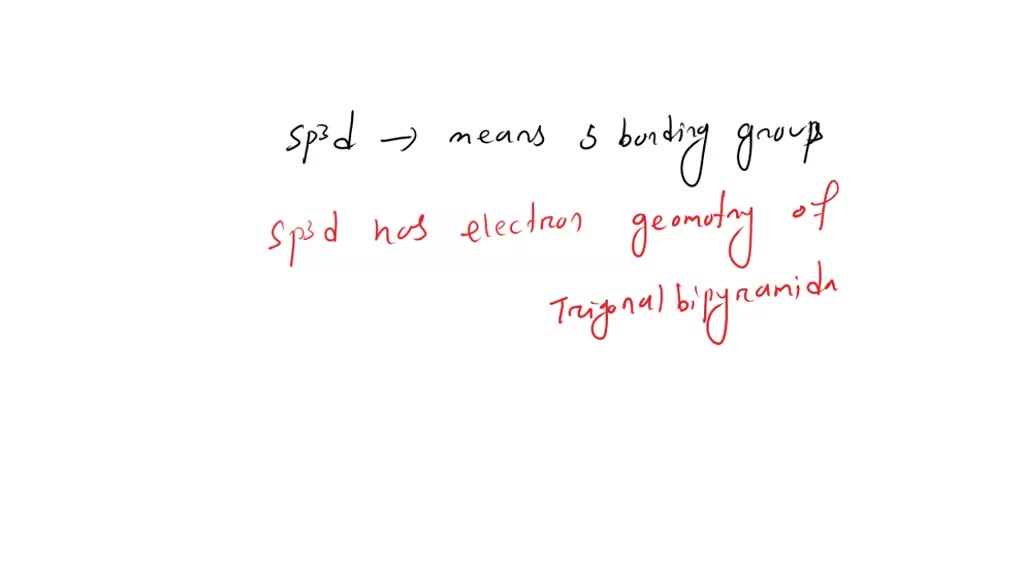A Molecule That Is Sp3d Hybridized And Has

Okay, let's talk about a molecule. You know, those tiny, invisible things that make up… well, everything! We're going to meet one that's a bit of a show-off in the world of atomic arrangements. It's like the molecule that decided to get a special haircut and wear mismatched socks.
This particular molecular friend is Sp3d hybridized. Now, I know that sounds like a secret code from a spy movie, but it just means its electrons are arranged in a rather fancy way. Think of it like having a super organized closet, but instead of shirts and pants, it's electrons doing a little dance.
And what does this Sp3d hybridization lead to? Well, it usually results in a molecule with a shape that’s a bit like a trigonal bipyramid. Picture a triangle with two little pointy hats on top and bottom. It's not your everyday, run-of-the-mill shape, is it?
Must Read
My unpopular opinion? I think these trigonal bipyramidal molecules are just misunderstood. They're the introverts at the molecular party, but they're secretly the most interesting ones if you just give them a chance. They’ve got these extra dimensions of electron positioning!
So, who is this intriguing molecule? One of the most common examples is Phosphorus pentachloride, or PCl5. Yes, phosphorus and five chlorine atoms decided to hang out together in this rather peculiar geometry.
When you look at PCl5, you can imagine the phosphorus atom in the center. It's like the popular kid everyone wants to be around, but instead of friends, it's got these chlorine atoms fanned out.
Three of the chlorine atoms are sort of doing a hula hoop dance around the equator. They're all in the same plane, like they're performing a synchronized swimming routine.
Then, you have the other two chlorine atoms. These guys are sticking straight up and straight down. They're like the enthusiastic cheerleaders, shouting from the top and bottom of the formation.

This arrangement gives PCl5 its unique trigonal bipyramidal shape. It’s a bit like a three-legged stool with a very tall hat and another very tall hat on the very top and bottom.
Now, I find this shape absolutely delightful. While most molecules are happy being linear (straight) or bent (like a grumpy cat) or even tetrahedral (like a tiny pyramid), PCl5 goes, "Nah, I need more angles." It's the molecule that refuses to be boring.
Think about it. You've got your everyday water molecule (H2O), all bent and cute. Then you've got methane (CH4), all tetrahedral and stable. They're nice, sure. Predictable, even.
But PCl5? It’s like showing up to a black-tie event in a sequined jumpsuit. It’s bold. It’s memorable. It’s… different.
And this difference is where the fun begins. Because of this specific electron arrangement, these Sp3d hybridized molecules have certain properties that make them super useful. It’s not just for show, you know!

For example, PCl5 is a fantastic reagent in organic chemistry. It loves to get rid of oxygen atoms. It’s like the ultimate declutterer of the molecular world.
Imagine you have a molecule that’s a bit too “oxygen-heavy.” PCl5 comes in, does its thing, and poof! The oxygen is gone, replaced by chlorine. It’s a bit of a chemical magic trick.
Chemists love PCl5 for this very reason. It helps them transform one molecule into another, creating all sorts of new and exciting compounds. It's like the Swiss Army knife of certain chemical reactions.
And it's not just PCl5! There are other molecules that also sport this Sp3d hybridization and the trigonal bipyramidal shape. For instance, Sulfur tetrafluoride (SF4) is another one. It’s a bit like PCl5 but with fluorine atoms instead of chlorine.
SF4 also has that intriguing shape. It’s sometimes described as a “seesaw” shape, which is kind of adorable. Imagine a seesaw with a sulfur atom in the middle and four fluorine atoms making it wobbly.

Then there's Iodine pentafluoride (IF5). This one is a bit more extreme. It’s got that trigonal bipyramidal framework, but it looks more like a square pyramid. It’s like the triangle hats on the PCl5 have grown a bit taller and wider.
These molecules might seem a little unusual, a little quirky. But that’s precisely what makes them so special. They break the mold. They dare to be different.
I just feel like in the grand scheme of molecular existence, the Sp3d hybridized ones deserve a little more appreciation. They’re not just awkwardly shaped; they’re elegantly shaped, in their own unique way.
It’s like the difference between a perfectly symmetrical, mass-produced piece of furniture and a handcrafted, slightly asymmetrical sculpture. Both have their place, but the sculpture often has more character, right?
So, the next time you hear about Sp3d hybridization, don't just think of complex electron theory. Think of PCl5, the molecule with the fancy haircut and the five chlorine friends doing a synchronized dance. Think of SF4, the seesaw molecule.

Think of them as the molecular rebels, the artists, the ones who didn't just settle for the standard shapes. They embraced the extra dimensions, the more complex angles, and in doing so, they became incredibly useful and endlessly fascinating.
It's a reminder that sometimes, the most interesting things are the ones that don't quite fit the typical mold. They might look a bit odd at first glance, but they often possess a hidden brilliance and a unique purpose.
So here’s to the Sp3d hybridized molecules! May they continue to surprise us with their geometry and their chemical prowess. They’re the unsung heroes of a more complex, and dare I say, more interesting molecular universe. And I, for one, am here for it!
It's like they're saying, "Why be linear when you can be a trigonal bipyramid?" And I think that's a beautiful sentiment.
So, while many might overlook them, I’ll be over here admiring their architectural ingenuity. They are, in my humble, popcorn-munching opinion, the coolest molecules on the block. The ones with the most… interesting personalities. They've clearly got more going on than just two or four arms!