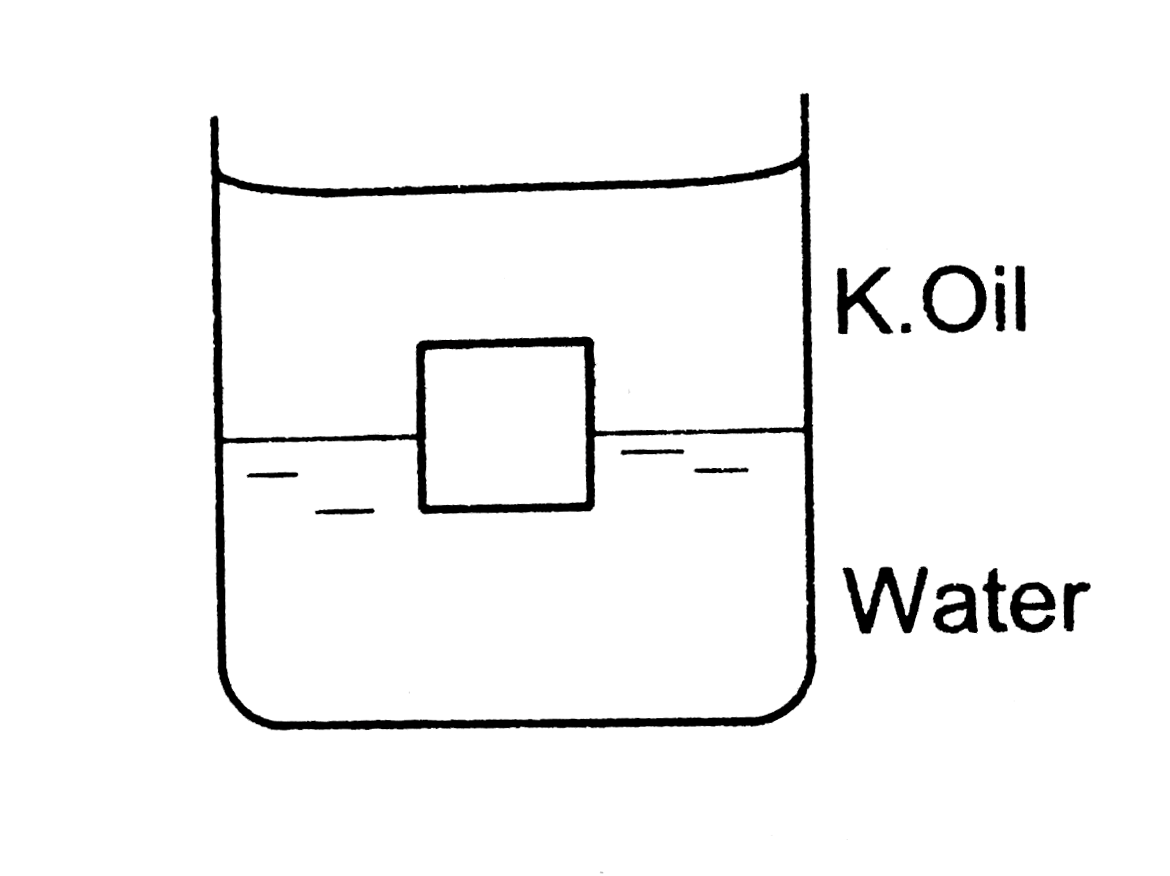
A Layer Of Oil Floats On An Unknown Liquid
Okay, so picture this: you’re staring into a clear container, maybe a fancy science-y beaker, or perhaps just a jam jar you “borrowed” from your mom’s pantry for an experiment (don’t tell her). And what do you see? Not just one liquid, oh no, this is a liquid situation. At the very top, shimmering like a misunderstood disco ball, is a layer of oil. Yep, regular old cooking oil, the stuff you use to make those sad, slightly burned fries.
But here’s the kicker, the plot twist that would make M. Night Shyamalan nod approvingly: underneath that oily sheen is… something else. A whole ‘nother liquid. And we have absolutely zero clue what it is. It’s like finding a mystery sock in your laundry, but instead of fuzzy and slightly alarming, it’s… liquid. And potentially even more alarming.
Now, you might be thinking, “Who cares about some random liquids?” And to that I say, my friend, you are missing out on the sheer, unadulterated weirdness of the universe. This isn't just a science demonstration; it’s a tiny, contained existential crisis. It's a visual metaphor for so many things: your relationship status (one person shining, the other completely undefined), your diet (sugar on top, regret underneath), or even just your Tuesday morning trying to assemble IKEA furniture. It’s a whole vibe.
Must Read
So, why does the oil behave like it owns the place, floating on top like a spoiled celebrity at a pool party? It all comes down to a little thing called density. Think of density as how much "stuff" is packed into a certain amount of space. Oil, bless its greasy heart, is less dense than most of the liquids it’s likely to hang out with. It’s basically the lightweight champion of the liquid world. It’s saying, “Nope, I’m too good for that heavy drama down below!”
Imagine you have a bag of feathers and a bag of rocks, both the same size. Which one is heavier? The rocks, right? Because they’re denser. They’ve got more rock-stuff crammed into that bag. Oil is like the feathers – light, airy, and happy to bob on the surface. The liquid underneath? It’s the rocks. It’s got more molecular oomph, more liquid-ness packed in there, so gravity says, “Yeah, you go down there, buddy.”

And this is where the mystery really kicks in. If we know oil is usually less dense than water, for example, then what is this unknown liquid lurking below? Is it water? It could be! But then why is the oil so dramatically on top? Perhaps the unknown liquid is something even less dense than water, but still denser than oil. My mind, as you can probably tell, is already racing. Is it some kind of super-concentrated lemonade? A potion brewed by a very confused witch? The tears of a clown who just stubbed his toe?
Let’s explore some possibilities, shall we? What if this unknown liquid is, in fact, water? Okay, normal. But then, the oil is floating on top. That’s expected. However, what if the water is so pure, so exceptionally pure, that it’s slightly less dense than your average kitchen oil? Unlikely, but not impossible. Science is full of “hold my beaker” moments. Or, perhaps, it's saltwater. Saltwater is denser than plain water. So, if our mystery liquid is just plain old water, and the oil is on top, that’s normal. But if it’s saltwater, then the oil is still on top? That means the oil is even less dense than saltwater. See? We’re getting into the nitty-gritty now.
But wait, there’s more! What if the unknown liquid is something a bit more… adventurous? What about alcohol? Like, say, a particularly strong vodka? Now we’re talking! Most common cooking oils are less dense than ethanol (that’s the fancy science word for alcohol). So, your oil could be chilling on top of a martini base. Cheers to that!
Or, brace yourselves, what if the unknown liquid is something like glycerin? Glycerin is significantly denser than water and oil. It’s thick, syrupy, and feels like you’re trying to stir a cloud. If our oil is floating on glycerin, that’s a pretty dramatic density difference. It’s like a tiny, oil-slicked raft on a sea of pure, viscous goo. Imagine trying to swim in that. You’d probably end up as a very shiny, very confused oil droplet.
The beauty of this situation, this layered liquid ballet, is that it teaches us about something called immiscibility. That’s a word that sounds way scarier than it is. It just means that two liquids don’t mix. Oil and water, for instance, are famously immiscible. They look at each other and go, “Nah, we’re good.” Oil and alcohol? They might get along a little better, but depending on the oil and the alcohol concentration, they can still keep their distance. It's like they have different social circles and refuse to mingle at the same molecular party.

So, when you see that layer of oil on an unknown liquid, you're witnessing a tiny, silent drama unfold. A drama of density, of molecular preferences, and of the fundamental laws of physics saying, "You go there, and you go over there." It's a miniature universe with its own rules, and we're just here, peering in, trying to decipher the liquid gossip.
And the funniest part? This isn't just happening in some sterile lab. This happens in nature! Think about oil spills. That oily sheen on the ocean? That’s the same principle at play. The oil is less dense than the saltwater, so it floats on top, causing all sorts of ecological chaos. It’s a stark reminder that sometimes, the most dramatic visual effects have very serious, real-world consequences. So, while you might be tempted to add some food coloring to your mystery jar to see what happens (don't do that unless you're prepared for staining!), remember that understanding these simple liquid behaviors can tell us a lot about the world around us.
So, the next time you encounter a layered liquid situation, whether it's in a science experiment, a spilled drink, or even just a particularly greasy pizza box, take a moment to appreciate the invisible forces at play. It's a little bit of magic, a little bit of science, and a whole lot of entertaining mystery. And who knows, maybe that unknown liquid is just waiting for its moment to shine… right underneath that glorious, shimmering layer of oil.


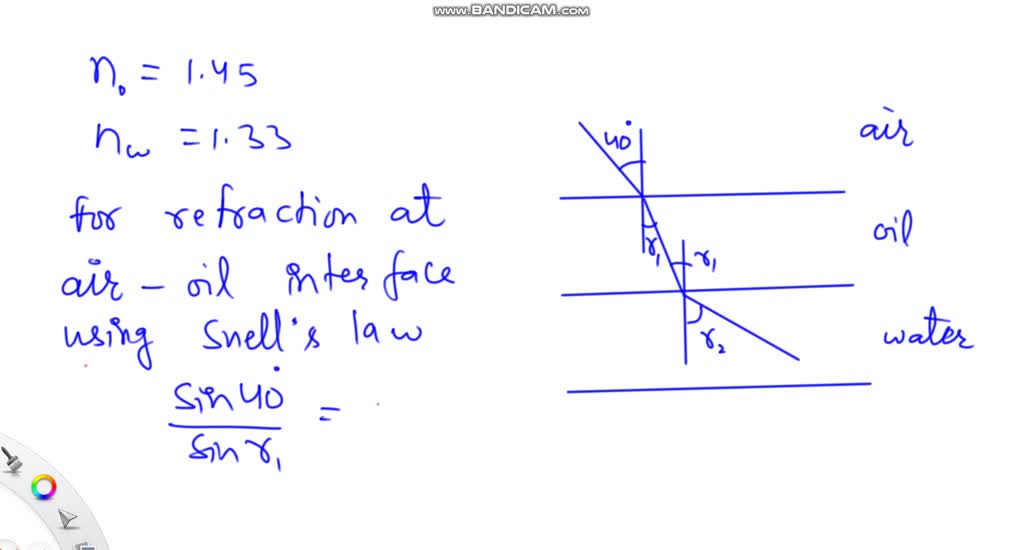
![SOLVED: 4.) [3 points] A tank holds a layer of oil (thickness To = 1](https://cdn.numerade.com/ask_images/f6cc4e8cfaf448d09ec9efd27af514bf.jpg)