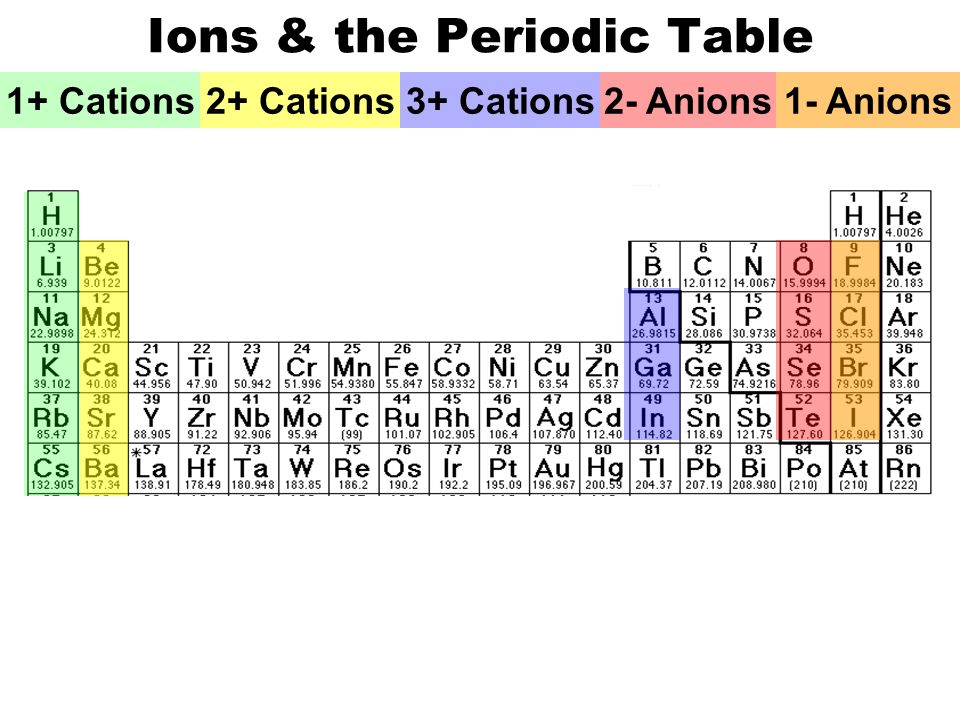
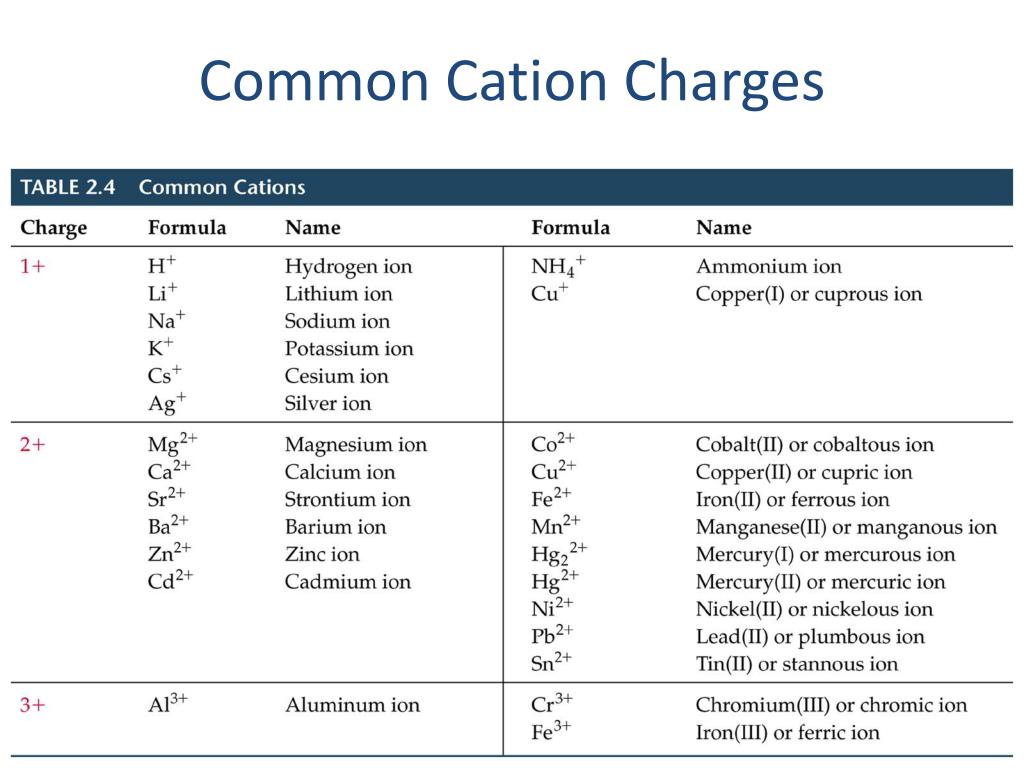
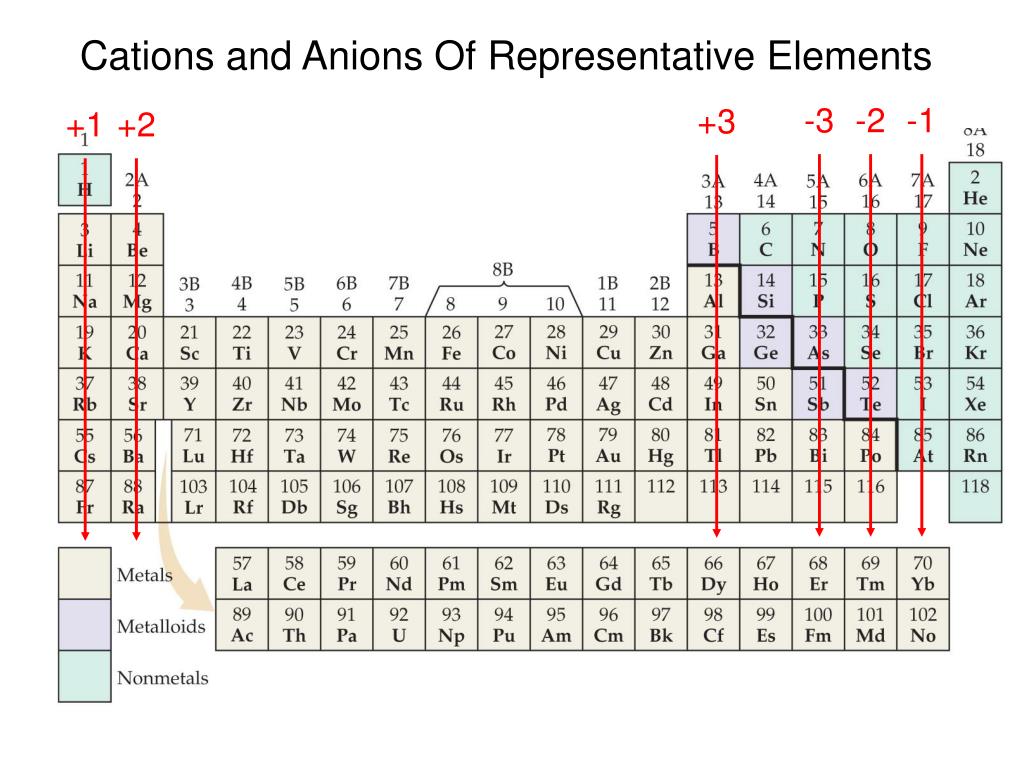
A Cation Of 2+ Indicates That An Element Has
Ever wondered what makes some elements in the periodic table so energetic, so eager to form bonds and create the amazing world around us? It all boils down to their "personality," and a big clue to that personality is whether they're sporting a "+" charge. Specifically, when we see a cation with a 2+ charge, it’s like spotting an element wearing a bright red cape, ready to jump into action! This isn't just some dry chemistry jargon; understanding these charged-up atoms unlocks the secrets behind everything from the vibrant colors in a fireworks display to the very structure of our bones and the electrical signals that power our brains.
Think of it this way: atoms are the building blocks of everything. They're usually quite content being neutral, with an equal number of positively charged protons in their core and negatively charged electrons buzzing around them. But sometimes, an atom feels like it has a little too much baggage. It’s holding onto more electrons than it really needs. So, what does it do? It bravely decides to donate two of its electrons to another atom that’s more than happy to take them. When an atom does this, it becomes positively charged because it now has more protons than electrons. And when it gives away two electrons, it proudly displays a charge of 2+. It's essentially saying, "I've got two extra negative charges I can share!"
The Superpowers of a 2+ Cation
This act of losing electrons isn't just a random chemical hiccup; it’s a strategic move that gives these elements special "superpowers." The most significant superpower is their enhanced reactivity. By shedding those two electrons, these atoms become highly attracted to other atoms that have an electron deficit or are looking to gain negative charges. This makes them fantastic partners in forming chemical bonds, the invisible glue that holds molecules together.
Must Read
One of the most compelling benefits of elements forming 2+ cations is their role in creating ionic compounds. Imagine a dance where one partner gives away two partners, and another partner eagerly accepts them. That’s essentially what happens. For example, the common table salt, sodium chloride (NaCl), is formed from a sodium atom (which typically forms a 1+ cation) and a chlorine atom. But many vital substances involve elements that form 2+ cations. Take calcium, for instance. Calcium readily forms a Ca2+ cation. This is crucial for our bodies! Your bones and teeth are made strong by calcium phosphate, where the calcium ions have that essential 2+ charge, interacting with phosphate ions. Without these 2+ calcium cations, our skeletal structures would be weak and brittle. It's a silent, constant marvel happening within us every single second!
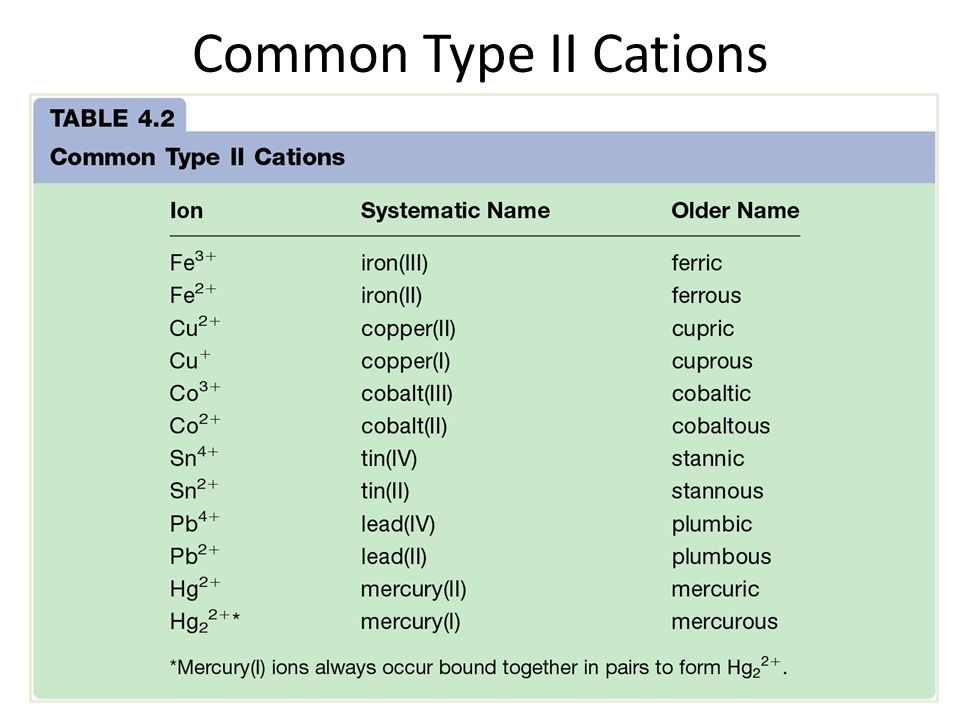
"The world is made of atoms, and when they gain or lose electrons, their 'attitude' completely changes, leading to fascinating chemical reactions."
Beyond biology, these 2+ cations are the unsung heroes of many everyday phenomena. Think about the dazzling colors in fireworks. Many of these brilliant hues are produced by metal salts, where metal ions with various charges, including 2+, are heated. For example, strontium salts, which contain strontium ions (Sr2+), are responsible for the vibrant reds in fireworks. Similarly, barium salts, featuring barium ions (Ba2+), contribute to the beautiful greens. These ions absorb energy when heated and then release it as specific wavelengths of light, creating the spectacular displays we enjoy.
From Batteries to Building Blocks
Let's dive a little deeper into another arena where 2+ cations are indispensable: electrochemistry. Batteries, the powerhouses of our modern lives, rely heavily on the movement of ions. In many common battery types, metals that form 2+ cations play a starring role. For instance, in a lead-acid battery (like the one in your car), lead atoms readily form Pb2+ ions. This ability to readily lose two electrons and participate in electrochemical reactions is what allows batteries to store and release electrical energy. It’s a direct consequence of their inherent tendency to become 2+ cations.
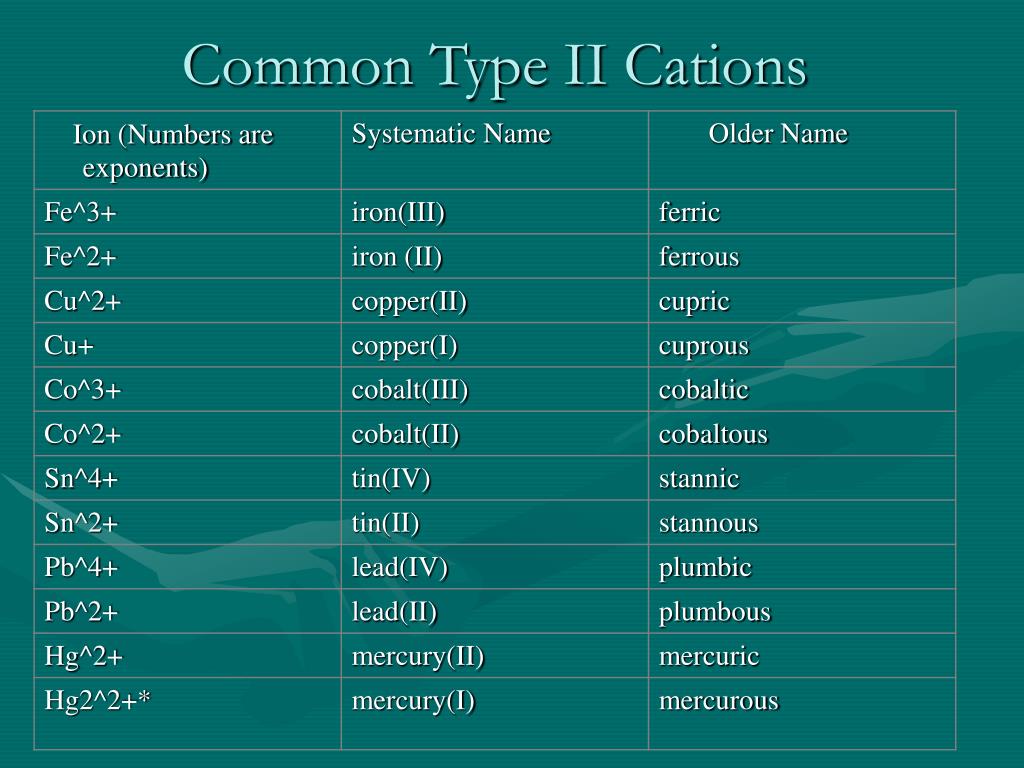
Even something as fundamental as rusting, the oxidation of iron, involves iron ions. While iron can form different charges, the formation of iron(II) ions (Fe2+) is a key step in this process, which ultimately leads to the familiar reddish-brown coating. Understanding this behavior helps scientists develop better methods for preventing corrosion and preserving the materials we rely on.
So, the next time you see a chemical formula or hear about elements interacting, remember the humble 2+ cation. It's not just a number and a symbol; it's a testament to an element's eagerness to connect, to build, to energize, and to create the vibrant, functional, and even explosive world we inhabit. It’s a fundamental aspect of chemistry that makes life, and indeed the universe, so wonderfully dynamic!